Why High-Purity Stainless Steel is Critical for Food and Pharmaceutical Safety

Table of Contents
What Makes Stainless Steel “Sanitary”?
 In industries where purity is paramount, such as food and pharmaceuticals, the materials used in production are critical.
In industries where purity is paramount, such as food and pharmaceuticals, the materials used in production are critical.
Contamination is not just a risk; it can be catastrophic. It directly impacts product integrity, consumer health, and brand trust. This is why high-purity stainless steel stands as a non-negotiable requirement. Its unique properties actively prevent unwanted interactions and ensure product safety.
Throughout this guide, we will explore what truly makes stainless steel “sanitary.” We will dig into its critical applications within sensitive environments. We will also cover the essential standards and regulations that guarantee safety and quality.
Join us as we uncover how high-purity stainless steel ensures the best corrosion resistance, superior cleanability, lasting durability, and ultimate product purity.
The term “sanitary”, when applied to stainless steel, goes far beyond mere cleanliness. It refers to a specific set of material properties and design characteristics that collectively ensure the highest levels of hygiene and product integrity in sensitive applications. At its core, sanitary stainless steel is engineered to prevent contamination, facilitate thorough cleaning, and resist degradation over time.
One of the most fundamental aspects is its inherent corrosion resistance. Unlike many other metals, stainless steel forms a passive chromium oxide layer on its surface when exposed to oxygen. This invisible, self-repairing film acts as a barrier, protecting the underlying steel from chemical attacks, rust, and pitting. In the aggressive environments of food and pharmaceutical processing, where strong acids, bases, and sanitizers are routinely used, this resistance is crucial. Without it, metals like copper can corrode, leach ions into the product, or create rough surfaces that harbor bacteria. While brass compression fittings for copper pipe offer some corrosion resistance for general plumbing, they cannot match the robust chemical inertness required for high-purity stainless steel systems.
Another key characteristic is its non-porous surface. The microstructure of sanitary stainless steel is extremely dense and smooth, lacking the microscopic pores and crevices found in other materials. This non-porous nature is vital because it denies microorganisms, product residues, and foreign particles places to accumulate and proliferate. A rough or porous surface would act as a breeding ground for bacteria, making effective cleaning impossible and leading to biofilm formation.
The surface smoothness is quantified by the Ra (Roughness Average) value. Lower Ra values indicate a smoother surface. For sanitary applications, extremely low Ra values are specified, often achieved through specialized polishing techniques. This smoothness is critical for preventing bacterial adhesion and ensuring that cleaning solutions can thoroughly rinse away all contaminants.
Furthermore, sanitary stainless steel components are designed with crevice-free construction. Every joint, seam, and connection point is carefully engineered to eliminate any gaps or dead spaces where product can stagnate or microbes can hide. This often involves seamless welding techniques and specialized fitting designs that present a continuous, uninterrupted surface to the product flow. This contrasts sharply with some traditional connections, like certain copper tube compression fittings, which inherently create small crevices at the ferrule-to-pipe interface.
Finally, all these properties contribute to Clean-in-Place (CIP) and Sterilize-in-Place (SIP) compatibility. The corrosion resistance, non-porous nature, smoothness, and crevice-free design ensure that processing equipment can be effectively cleaned and sterilized without disassembly. This reduces downtime, minimizes human error, and guarantees a consistently hygienic environment.

Common Grades of Sanitary Stainless Steel
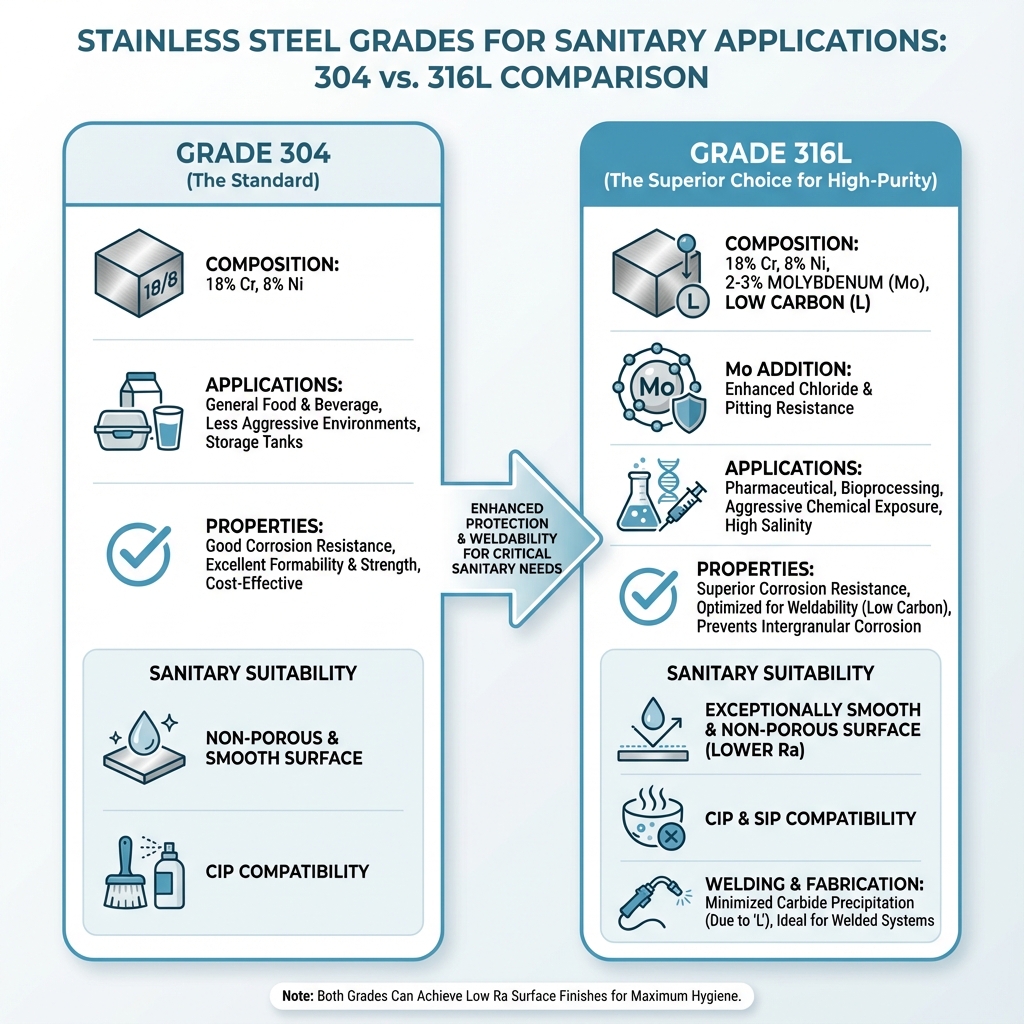
While various types of stainless steel exist, specific grades are predominantly used in sanitary applications due to their superior properties. The most common among these are Grades 304 and 316, particularly Grade 316L. Understanding their differences is key to selecting the appropriate material for a given application.
Grade 304 Stainless Steel is the most widely used austenitic stainless steel. It contains approximately 18% chromium and 8% nickel, which gives it excellent corrosion resistance, good formability, and high strength. It is suitable for many food and beverage applications where corrosive conditions are not excessively harsh. Its widespread availability and cost-effectiveness make it a popular choice for general-purpose sanitary equipment, such as storage tanks and work surfaces, and for less aggressive processing lines.
Grade 316 Stainless Steel builds upon the properties of 304 by adding molybdenum (typically 2-3%). This addition significantly improves its corrosion resistance, especially against chlorides and other halogen compounds, which are often present in cleaning agents, brines, and some food products. This makes 316 a preferred choice for environments with higher salinity or more aggressive chemical exposure.
Grade 316L Stainless Steel is a low-carbon version of 316. The “L” stands for “low carbon,” meaning its carbon content is limited to 0.03%. This reduced carbon content is crucial for welding applications. When stainless steel is welded, the heat can cause chromium carbides to precipitate at the grain boundaries, a phenomenon known as “sensitization.” This depletes chromium from the surrounding areas, making the steel susceptible to intergranular corrosion. By reducing its carbon content, 316L minimizes carbide precipitation, thereby preserving its corrosion resistance in the heat-affected zones of welds. This makes 316L the material of choice for most high-purity and sanitary applications that require extensive welding, such as pharmaceutical processing, bioprocessing, and certain dairy equipment.
The molybdenum addition in 316 and 316L is particularly noteworthy. It provides improved resistance to pitting and crevice corrosion, common forms of localized attack that can compromise equipment integrity and cleanability. This is a critical advantage over materials like copper, which can be susceptible to pitting in certain water chemistries, potentially leading to leaks or contamination.
To illustrate the differences, we can compare these grades:
The Importance of Surface Finish
Beyond the chemical composition, the physical state of the stainless steel surface, known as its surface finish, is a paramount factor in sanitary design. The surface finish directly impacts the material’s cleanability, resistance to bacterial adhesion, and overall hygienic performance.
The primary metric for measuring surface roughness is Ra (Roughness Average). A lower Ra value indicates a smoother surface. In sanitary applications, we strive for extremely low Ra values, typically measured in microinches (µin) or micrometers (µm). For example, pharmaceutical and bioprocessing equipment often specifies internal surface finishes of 20 Ra µin (0.5 Ra µm) or even smoother, down to 10 Ra µin (0.25 Ra µm) for critical product contact surfaces.
Achieving these ultra-smooth finishes involves specialized processes:
- Mechanical Polishing: This involves progressively finer abrasives to grind and buff the surface. While effective, it can leave microscopic scratches and residual abrasive particles if not performed carefully.
- Electropolishing: An electrochemical process that selectively removes a thin layer of material from the stainless steel surface, preferentially dissolving peaks and leaving valleys. The result is an exceptionally smooth, bright, and passive surface. Electropolishing further reduces the Ra value, improves corrosion resistance by increasing the chromium-to-iron surface ratio, and effectively removes microscopic imperfections that could harbor bacteria.
The direct benefit of a superior surface finish is the prevention of biofilm formation and bacterial adhesion. Biofilms are communities of microorganisms encased in a self-produced polymeric matrix, which can adhere tenaciously to surfaces. Once formed, biofilms are extremely difficult to remove, even with aggressive cleaning and sanitization. A rough surface provides numerous microscopic nooks and crannies where bacteria can attach and begin forming a biofilm. A smooth, electropolished surface, conversely, offers minimal attachment sites, making it much harder for bacteria to colonize and significantly easier for CIP/SIP processes to dislodge any transient microorganisms. This is a crucial distinction when choosing materials for critical processes, where even minute surface imperfections can compromise product safety.
Critical Applications in High-Purity Industries
The demand for high-purity stainless steel is driven by the stringent requirements of industries where product integrity, consumer safety, and regulatory compliance are non-negotiable. These sectors cannot afford any compromise in material quality, as even trace contamination can have severe consequences.
In food and beverage processing, stainless steel is ubiquitous. From dairy farms to breweries, and from juice production to confectionery, it is the material of choice for tanks, pipelines, heat exchangers, and processing equipment. Its resistance to food acids and sanitizers, and its non-reactive nature, ensure that the taste, purity, and safety of consumables are maintained. Consider a modern brewery: the immense stainless steel fermentation tanks, intricate piping systems, and precise valves are all designed to prevent microbial spoilage and maintain the quality of the brew.

Pharmaceutical manufacturing represents perhaps the pinnacle of high-purity requirements. Here, stainless steel is used for everything from active pharmaceutical ingredient (API) synthesis reactors to sterile water distribution systems and final product formulation tanks. The absolute necessity of preventing cross-contamination, ensuring product stability, and meeting strict regulatory guidelines (such as FDA cGMP) means that only the highest grades of electropolished 316L stainless steel are acceptable. The slightest material degradation or microbial ingress could render an entire batch of life-saving medication unusable or, worse, harmful.
Bioprocessing, which involves the production of biological materials like vaccines, antibodies, and enzymes, also relies heavily on high-purity stainless steel. These processes often involve delicate cell cultures and highly sensitive biological molecules that are easily affected by contaminants. The equipment must be capable of maintaining sterility throughout complex fermentation, purification, and filtration steps.
The dairy industry is another prime example. Milk and dairy products are highly susceptible to bacterial growth. Stainless steel’s ease of cleaning, resistance to lactic acids, and ability to withstand frequent sanitization cycles make it indispensable for processing, storage, and transportation of dairy products, safeguarding public health.
Sanitary Stainless Steel in Processing Equipment
The versatility and superior properties of high-purity stainless steel make it the material of choice for a wide range of components in processing equipment for these critical industries. Every part that comes into contact with the product must adhere to the highest sanitary standards.
Tanks and vessels are perhaps the most visible applications. From large fermentation tanks in breweries to small mixing vessels in pharmaceutical labs, these containers are typically constructed from 304 or 316L stainless steel, often with electropolished internal surfaces. Their seamless construction and robust design ensure product containment and prevent external contamination.
Piping and tubing systems form the arteries of any processing plant. High-purity stainless steel tubing, often orbitally welded, transports raw materials, intermediates, and finished products. Unlike general plumbing applications, which might use copper tube compression fittings for water lines, high-purity industries demand stainless steel throughout their piping and tubing systems. This ensures chemical compatibility, prevents leaching, and facilitates thorough CIP cleaning. The smooth internal bore of stainless steel tubing minimizes friction and prevents product hang-up, further contributing to hygiene.
Valves and pumps are critical control points in any fluid handling system. Sanitary valves (e.g., diaphragm, ball, butterfly valves) and pumps (e.g., centrifugal, positive displacement) are specifically designed from stainless steel to be easily cleanable, crevice-free, and to prevent product entrapment. Their internal surfaces are polished to match the piping, maintaining a continuous sanitary pathway.
Heat exchangers are essential for temperature control in many processes. Plate-and-frame or shell-and-tube heat exchangers constructed from stainless steel ensure efficient heat transfer without contaminating the product. The material’s corrosion resistance is particularly important here, as temperature fluctuations can exacerbate corrosive tendencies.
Essentially, any product-contact surface in these industries must be made of high-purity stainless steel. This includes agitators, filters, nozzles, and even small components like sample ports. The consistent use of this material across all these elements creates a cohesive, hygienic processing environment.
Why Component Quality Matters
The integrity of a high-purity system is only as strong as its weakest link. This principle underscores the critical importance of every single component, from the largest vessel to the smallest fitting. Compromising on the quality of any part can undermine the entire system’s sanitary performance and lead to catastrophic failures in product safety.
Fittings and connectors are particularly vulnerable points if not chosen and installed correctly. While some general industrial applications might rely on various materials for compression fittings, including copper or brass, high-purity environments demand stainless steel fittings that match the piping in material grade and surface finish. These fittings must be designed to be crevice-free, allowing for smooth product flow and effective cleaning. Types of sanitary fittings include tri-clamp, butt-weld, and flanged connections, each chosen for its specific advantages in maintaining hygiene and ease of disassembly for inspection if required. Even in systems where some components might traditionally use brass or copper compression fittings, the critical nature of food and pharmaceutical production necessitates the superior corrosion resistance and hygienic properties of stainless steel for all contact surfaces.
Weld integrity is another non-negotiable aspect. In high-purity systems, welding is often preferred over mechanical connections to create seamless, crevice-free joints. However, a poorly executed weld can introduce porosity, cracks, or surface imperfections that become breeding grounds for bacteria. This is why specialized orbital welding techniques, performed in controlled environments with inert gas purging, are standard practice. These welds are then often electropolished to ensure a smooth, passive surface.
Gasket materials also play a crucial role. While not stainless steel themselves, gaskets must be compatible with the process fluids, cleaning agents, and temperatures. They must be made from FDA-approved materials (e.g., EPDM, PTFE, Viton) that are non-shedding, non-reactive, and designed to create a tight, hygienic seal without extruding into the product stream.
Sourcing top-quality sanitary stainless steel components from reputable suppliers is paramount. This ensures that all parts, from tubing to fittings and valves, meet the exacting material specifications, surface finish requirements, and design standards necessary for high-purity applications. When every component is designed and manufactured to the highest sanitary standards, the overall system reliability is dramatically improved. This translates to consistent product quality, reduced risk of contamination, minimized downtime for cleaning and maintenance, and ultimately, safeguarding consumer health and brand reputation.
Key Standards and Regulations for Sanitary Stainless Steel
In the high-purity industry, relying solely on manufacturers’ claims is insufficient. A robust framework of standards and regulations defines, certifies, and enforces requirements for sanitary equipment and materials. These guidelines are crucial for ensuring safety and quality and for providing a common language for design, fabrication, and operation across the globe. They serve as benchmarks that manufacturers must meet, and that end-users can trust.
These standards are typically developed by industry associations and regulatory bodies, often incorporating input from engineers, scientists, and public health experts. They cover a wide range of aspects, from material composition and surface finish to design principles that facilitate cleaning and prevent bacterial growth. While general plumbing standards for copper tube compression fittings focus on pressure integrity and material compatibility, sanitary standards go far beyond, emphasizing biological and chemical inertness and cleanability.
Understanding 3-A Sanitary Standards
Among the most recognized and respected guidelines in the food, beverage, and dairy industries are the 3-A Sanitary Standards. Developed by 3-A Sanitary Standards, Inc., a non-profit organization, these standards provide detailed hygienic design criteria for equipment and processing systems. The “3-A” originates from the three founding organizations: the American Dairy Science Association, the U.S. Public Health Service, and the Dairy Industry Committee.
The core purpose of 3-A standards is to protect public health by ensuring that food and dairy equipment is designed to be easily cleanable, preventing product contamination and promoting food safety. Key aspects covered by 3-A include:
- Material Requirements: Only specific materials, primarily stainless steel grades like 304 and 316L, are approved, along with certain plastics and elastomers that are non-toxic, corrosion-resistant, and non-absorbent.
- Hygienic Design Criteria: Equipment must be designed to be smooth, non-porous, and free of sharp angles, dead ends, and crevices where microorganisms can accumulate. Internal radii must be sufficiently large to allow for thorough cleaning.
- Fabrication Standards: Welding must be smooth, continuous, and free of pits or cracks. Joints must be flush.
- Cleanability: Designs must facilitate effective Clean-in-Place (CIP) or manual cleaning, ensuring all product contact surfaces can be reached and cleaned.
- Disassembly: Equipment that requires manual cleaning or inspection must be easily and quickly disassembled and reassembled without specialized tools.
Equipment that meets these rigorous standards can bear the 3-A Symbol, which is a widely recognized mark of hygienic design and construction. This is a voluntary certification, but processors and regulators highly value it as it signifies a commitment to the highest levels of food safety.
The Role of ASME-BPE
For the pharmaceutical and bioprocessing industries, the ASME Bioprocessing Equipment (BPE) standard is the definitive guide. Published by the American Society of Mechanical Engineers (ASME), the BPE standard provides comprehensive requirements for the design, materials, fabrication, inspection, and testing of equipment used in the manufacture of biopharmaceuticals.
The ASME-BPE standard is specifically designed to ensure sterility and prevent contamination in highly sensitive biopharmaceutical processes. It addresses unique challenges such as microbial control, cross-contamination, and the need for absolute product purity. Its scope is incredibly detailed, covering:
- Materials: Specifies acceptable grades of stainless steel (predominantly 316L), surface finishes, and non-metallic materials, along with their testing and certification requirements.
- Design for Sterility: Mandates designs that minimize dead legs, promote drainability, and facilitate complete sterilization (SIP).
- Material Joining: Provides extensive guidelines for orbital welding, weld quality, and post-weld treatment to ensure smooth, crevice-free, corrosion-resistant joints. This is a significant departure from mechanical connections, such as copper tube compression fittings, which are typically not suitable for sterile environments governed by ASME-BPE.
- Surface Finish Specifications: Sets extremely stringent requirements for internal and external surface finishes, often specifying electropolishing and precise Ra values to prevent microbial adhesion and facilitate cleaning.
- Testing and Inspection: Outlines procedures for verifying the integrity and cleanliness of equipment, including passivation, leak testing, and surface finish measurement.
Adherence to ASME-BPE is critical for biopharmaceutical manufacturers seeking to comply with regulatory requirements from bodies like the FDA. It ensures that equipment is not only robust and reliable but also inherently designed to produce safe, pure, and effective biopharmaceutical products.
Frequently Asked Questions about High-Purity Steel
We often encounter common questions regarding the selection, maintenance, and application of high-purity stainless steel. Addressing these helps clarify its critical role in sensitive industries.
What is the main difference between 304 and 316 sanitary stainless steel?
The primary distinction between 304 and 316 sanitary stainless steel lies in their composition: 316 contains molybdenum. Grade 304 contains approximately 18% chromium and 8% nickel. Grade 316, on the other hand, adds 2-3% molybdenum to this mix.
This molybdenum addition is crucial for enhancing chloride resistance. Chlorides, common in many food products (like brines), cleaning agents, and pharmaceutical formulations, can cause pitting and crevice corrosion in 304 stainless steel. The molybdenum in 316 provides superior protection against localized corrosion, making it more suitable for harsher, more corrosive environments.
In terms of cost vs. performance, 304 is generally more economical and perfectly adequate for many applications with minimal or moderate chloride exposure. However, for applications involving high chloride concentrations, aggressive chemicals, or liftd temperatures, the increased corrosion resistance of 316 (or 316L for welding) justifies its higher cost. Therefore, the suitability of the application is the deciding factor. While 304 might be used for general storage tanks, 316L is typically mandated for pharmaceutical reactors or marine environments where chloride attack is a significant concern.
How is sanitary stainless steel cleaned and passivated?
Maintaining the sanitary properties of stainless steel requires rigorous cleaning and passivation.
Cleaning typically occurs through Clean-in-Place (CIP) and Sterilization-in-Place (SIP) systems. CIP involves circulating various cleaning agents (e.g., alkaline detergents to remove organic soils, acidic detergents to remove mineral deposits, and sanitizers to kill microorganisms) through the equipment without disassembly. The smooth, non-porous surface of sanitary stainless steel ensures these agents can effectively remove all residues. SIP then uses steam or hot water to sterilize the system.
After fabrication or whenever the passive layer has been compromised (e.g., by welding, mechanical damage, or exposure to harsh chemicals), passivation is performed. This passivation process is a chemical treatment that restores and improves the chromium oxide passive layer on the stainless steel surface. The most common methods involve exposing the surface to nitric acid or citric acid solutions. These acids selectively remove free iron from the surface, allowing the chromium to react with oxygen and restore the chromium oxide layer. This process is critical for maximizing corrosion resistance and preventing surface rust, which could otherwise lead to contamination. Unlike simply cleaning, passivation is about restoring the material’s inherent protective properties.
Can you weld sanitary stainless steel components?
Yes, sanitary stainless steel components are routinely welded, and welding is often the preferred method for creating seamless, crevice-free connections in high-purity systems. However, it requires specialized techniques to maintain sanitary properties.
The most common method is orbital welding, a mechanized Tungsten Inert Gas (TIG) welding process. Orbital welding ensures consistent, high-quality welds that are smooth, uniform, and free from defects. This method is preferred because it minimizes human error and produces repeatable results.
A critical aspect of welding sanitary stainless steel is purging with inert gas, typically argon or a mixture of argon and helium. This involves filling the inside of the pipe or component with inert gas during welding. The purpose is to prevent the molten metal from reacting with oxygen in the air, which would lead to oxidation (sugaring or discoloration) on the inside surface of the weld. Such oxidation creates a rough, porous surface that is prone to corrosion and bacterial adhesion, completely compromising the sanitary integrity. By preventing oxidation, purging ensures that the internal weld bead is as smooth and clean as the parent material.
Proper welding techniques are essential for maintaining sanitary properties. While simpler connections, such as copper tube compression fittings, are often installed mechanically, high-purity stainless steel systems usually rely on orbital welding to create seamless, crevice-free joints that maintain the highest levels of hygiene and integrity. Post-weld treatments, such as electropolishing or passivation, are often applied to improve further the surface finish and corrosion resistance of the welded areas, ensuring a continuous, hygienic surface throughout the system.
Conclusion
In the demanding landscapes of food, pharmaceutical, and bioprocessing industries, the choice of materials is never a trivial decision. As we have explored, high-purity stainless steel is not merely a preference; it is a fundamental and non-negotiable material requirement. Its unparalleled corrosion resistance, non-porous and ultra-smooth surfaces, and inherent compatibility with rigorous cleaning and sterilization protocols make it the only viable option for safeguarding product integrity and consumer health.
From the molecular structure that resists chemical attack to the carefully polished surfaces that thwart microbial adhesion, every characteristic of sanitary stainless steel is engineered to uphold the highest safety standards. We have seen how specific grades, such as 316L, combined with advanced fabrication techniques and stringent surface finish requirements, form the backbone of hygienic processing environments. Furthermore, adherence to comprehensive guidelines like 3-A Sanitary Standards and ASME-BPE ensures that every component, from tanks to the most intricate fittings, meets global benchmarks for design, performance, and cleanability.
As industries continue to evolve, driven by innovation and ever-increasing regulatory scrutiny, the future of sanitary design will undoubtedly continue to center on advanced stainless steel solutions. The ongoing development of new alloys, surface treatments, and fabrication methods will only further improve its capabilities. Our collective commitment to precision, quality, and hygiene in these critical sectors underscores the imperative to prioritize quality in every material selection. By doing so, we not only protect the products we consume and rely upon but also reinforce the trust that consumers place in these vital industries.






